ʻO keʻano nui ma waena o nā nitrite a me nitrite e like me nā mea e like me nā oxygen i hoʻopaʻaʻia i kahi atom o Nitrogen i kahi o Nitrogenʻelua i hoʻopaʻaʻia i kahi atom
ʻO nā niterate a me Nitrite he mau'āpana i loko o nā'āpana o nā nitrogen a me nā oxygen oxygen. Loaʻa i kēia mau'āpanaʻelua i kahi uku uila. Hele mai lākou e like me ke aniona o nā mea paʻakai paʻakai. Aia kekahi mauʻokoʻa ma waena o ka nitrate a me nitrite; E kūkākūkā mākou i kēlā mauʻokoʻa i kēiaʻatikala.
He aha ka nitrate?
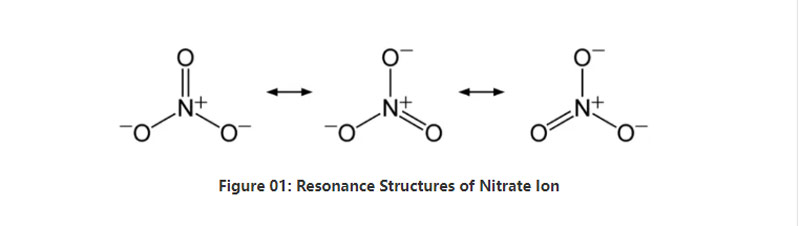
ʻO ka nitrate kahi aniyorganic ani e loaʻa ana i keʻano o keʻano chemula no3-. He aniyomomic anon he 4 atom; hoʻokahi nitrogen atom a meʻekolu oxygen i nā atom. ʻO ka anion he uku nui loa. ʻO ka hapa nui o ka molar o kēia anion he 62 g / mol. Eia kekahi, ua loaʻa kēia aniona mai kāna acid conjugate; nitric acid or hno3. Ma nā hua'ōlelo'ē aʻe,ʻo nitrate ka kumu o ke kumu o ka nitric acid.
I loko o ka pōkole,ʻo Nitrate Ion i hoʻokahi nitrogen atom ma ke kikowaena e pili ana i nā mea e pili ana i nā atom o ka nui. Ke noʻonoʻo nei i keʻano o keʻano o kēiaʻano, heʻekolu mau mea kūponoʻole (e like me keʻano o ka hoʻoponopono o ka anino). No laila,ʻo ka geometry o ka molecule he hoʻolālā trigonal. ʻO kēlā me kēia oxygen ATOM e lawe ana i kahi hana - 2/3 mau kālā, e hāʻawi ana i ka uku holoʻokoʻa o ka aniho e like me -1.
Ma ka palena maʻamau a me ka mahana, kokoke i nā pūpū paʻakai a pau e loaʻa ana i kēia anino i hōʻeha i ka wai. Hiki iā mākou ke loaʻa maoli i ka hanaʻana i nā mea nitrarate i ka honua ma ka honua e like me nā waihoʻia; nā waihona nitratine. ʻO ia ka mea nui loa i ka sodium nitrate. Eia kekahi, hiki i ka mea hānai ke kiʻi i nā balena ke hana i ka nitrate ion. ʻO kekahi o nā hoʻohana nui o nā mea hoʻohana o nā balts i loko o ka hanaʻana o nā certerlizers. Eia nō naʻe, pono ia e like me ka mea i hoʻonuiʻia ma luna o nā mea hana i nā mea hana.
He aha ka nitrite?
ʻO Nitrite kahi paʻakai ma loko o ka paʻakai i loaʻa i keʻano o ke kemika no2-. ʻO kēia anion he symmetric anion, a loaʻa iā ia kahi atom nitrogen atom i hoʻopaʻaʻia iʻelua oxygen a meʻelua mau mea paʻa. No laila,ʻo ka atom i ke kikowaena o ka molena. ʻO ka anion he uku nui loa.
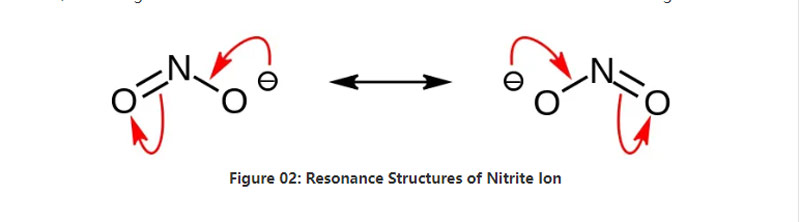
ʻO ka nui o ka molar o ke anionina he 46.01 g / mol. Eia nō naʻe, ua loaʻa i kēia anion i kahi o ka nitfous acid a iʻole hno2. No laila, he kumu kumuwaiwai o ka nidrous. No laila, hiki iā mākou ke hana i nā paʻakai nitrite ma Via e hala ana i nā hana koisa i ka sodium hydroxidel. Eia hoʻi, hana kēia i ka sodium nohua eʻike mākou ma keʻano o ka mea hou. Eia kekahi,ʻo nā paʻakai nittite e like me keʻano o ka sodium nitrite no ka mālamaʻana i ka meaʻai no ka mea hiki ke pale i ka meaʻai mai ka uluʻana o ka microbial.
He aha kaʻokoʻa ma waena o ka nitrate a me nitrite?
ʻO ka nitrate kahi aniyorganic ani e loaʻa ana i keʻano chemula no3- No laila,ʻokoʻa kaʻokoʻa mua ma waena o ka nitrate a me nitrite ma luna o keʻano o keʻano o nā'āpanaʻelua. ʻo ia; ʻO keʻano nui ma waena o nā nitrite a me nitrite e like me nā mea e like me nā oxygen i hoʻopaʻaʻia i kahi atom o Nitrogen i kahi o Nitrogenʻelua i hoʻopaʻaʻia i kahi atom Eia nō hoʻi, loaʻa ke kiʻiʻia mai o ia i ka acika hana pona; ʻO ka nitric acid,ʻoiaiʻo ka nitrite i loko o ka nitrite i loaʻa mai i ka nittrous acid. E like me kahiʻokoʻa'ē aʻe ma waena o nā nitrite a me nā nitrite ions, hiki iā mākou ke'ōlelo i keʻano no ka mea e hoʻokaʻawale i kahi o nā nitrite a me ka hoʻemiʻana o ka nitrite a hōʻemi i nā mea hana maʻamau.
POST manawa: Mei-16-2022





